利用Mastercycler X50 PCR仪与epMotion系统实现高重复性的微...
利用Mastercycler X50 PCR仪与epMotion系统实现高重复性的微量体积PCR实验
Highly reproducible low Volume PCR with Mastercycler® X50 and epMotion®
Arora Phang, Tim Schommartz, Eppendorf AG, Hamburg, Germany
Abstract
Key requirements of a thermal cycler are naturally accuracy and
reproducibility in terms of temperature performance. However, due to
increasingly robust PCR reagent formulations in the market, temperature
performance of a thermal cycler can be difcult to determine until one
comes across a delicate PCR system. Optimization of such a system then
becomes very tedious, laborious and can be extremely resource-consuming.
Poor temperature performance of a thermal cycler and evaporation are
two common issues that lead to poor PCR results.
However, these two factors are difcult to simply assess based on
numbers on a technical specifcation sheet. This paper demonstrates how
Eppendorf epMotion effortlessly handles low volume PCR set-up, hence
cutting down preparation effort while simultaneously increasing accuracy.
Furthermore, it presents the robustness of the new Eppendorf
Mastercycler X50 even when running low volume PCR, demonstrating its
excellent temperature performance and protection against evaporation.
Introduction
Modern scientifc trends tend towards higher speed, greater convenience
and lower volumes. Similarly, scientifc equip ment follows through this
line of evolution to push such lim its, giving rise to fancier
equipment and techniques. Though easily overshadowed in the wake of such
exciting advance ment, performance breakthroughs in simple and
established
equipment can also be more useful as they are generally less resource intensive to operate.
Large PCR setups can be very time-consuming and tedious if done
manually. Especially when handling very small volumes; imprecision and
pipetting errors caused by the operator can lead to massive variations
of the results. This operator-based error carries an even bigger
implication when different individuals are involved in the same work flow.
A way to overcome this issue is to use an automated liquid handling
platform. This allows an increase of sample throughput while at the same
time ensuring a reliable handling of small volumes with minimal
variations between experimental runs.
Temperature performance of a thermal cycler is generally measured
using an external temperature verifcation device. Such a measurement
method can provide a good indication on the performance of the thermal
block of a cycler. However, overall PCR performance relies on not just
the thermal block but also the additional combination of consumable
compatibility and ability to maintain reaction concentration by
protecting against evaporation. Thus, the results of an actual PCR run
using a temperature sensitive PCR system would present the best direct
empirical proof of reliable performance of a thermal cycler.
Additionally, a thermal cycler performance in terms of accuracy,
homogeneity and robustness can be verifed by performing the same PCR
under stricter conditions such as lower reaction volumes, enzyme
concentration and template quantity. The results from this Application
Note will illustrate the convenience of the epMotion and robustness of
the Mastercycler X50 in handling low volume PCR setup and runs.
Materials and Methods
PCRBio Taq DNA polymerase (NIPPON Genetics) and Human Genomic DNA
(Roche®) were used for the following amplifcation. PCR reaction master
mix was prepared with 1X reaction buffer and 0.2 µM of each primer, for
fnal volumes of 3 µL or 10 µL per reaction respectively. For 10µL
reaction, 0.25U of enzyme and 20 ng DNA were used while when amplifying 3
µL reaction volume, the amount of enzyme and DNA was reduced to 0.15U
and 10 ng, respectively. The master mix was dispensed into all 96 wells
of Eppendorf twin.tec® skirted PCR plates.
The following primers were used for amplifcation of the human ß-actin gene:
Forward primer: 5’- ATCGCCGCGCTCGTCGTC-3’
Reverse primer: 5’- TGGGTCATCTTCTCGCGGTTGG-3’
Dispensing was carried out by Eppendorf epMotion 5073l equipped with a 0.2–10 µL TS10 single channel dispensing tool using free jet dispensing in pipetting mode. The work table-setup is displayed in Figure 1. Furthermore, this dis pensing protocol uses a device-default template and hence can be easily adapted for other Eppendorf epMotion models equipped with a 0.2–10 uL dispensing tool.
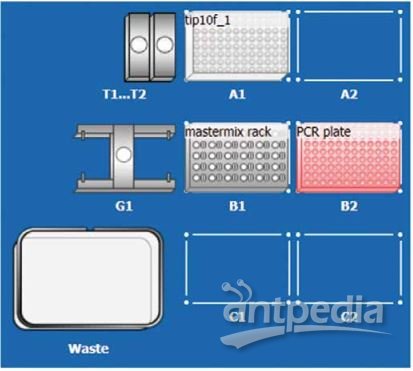
Figure 1: epMotion 5073l worktable for PCR setup
Plates were sealed with Eppendorf
Adhesive PCR Film and PCR were carried out on Mastercycler X50s. Cycling
conditions are listed in Table 1. The PCR products were detected using
GelRedTM (Biotium) following agarose gel electrophoresis and visualized
using the Gel Doc XR+ (BioRad®).
Table 1: PCR condition with two concurrent gradient setting at denaturation and annealing steps.
Results and Discussion
The new Eppendorf epMotion TS10 pipetting tool and Mastercycler X50
are a powerful combination in running low volume applications. Not only
does an automated pipetting system provide superior accuracy, it is also
comparatively more pleasant than setting up PCR reactions manually,
especially at high throughput. The error tolerance range is naturally
narrower when working with lower liquid volumes. Therefore, automated
systems with defned and controlled
force provide more reproducible dispensing of PCR reagents, which in
turn result in higher confdence in PCR results. The use of an automated
liquid handler also allows the operator to reduce hands-on setting-up
time to a bare minimum.
Additionally, evaporation issues in a thermal cycler are intensifed at
lower reaction volumes due to the lower amount of water present in the
mastermix. Hence, a change in even a small amount of liquid will lead to
a big shift in reagent concentration that will affect PCR amplifcation.
As evaporation in each well across the block is uncontrollable,
irreproducibility is the most common consequence of evaporation.
Therefore, a good thermal cycler is one that has good thermal
performance as well as a design that protects against evaporation.

These evaluation criteria can be easily
tested using a temperature sensitive PCR system. The human ß-actin gene
used in this study is one such system. Specifc amplifcation will yield
484 bp fragments while sub-optimal condition will give rise to
non-specifc amplifcation visible as a 350 bp artefact in the gel. This
characteristic can be used to clearly portray how fnely temperature
control a cycler has. Figure 2A showed the PCR amplifcation of the
ß-actin gene across different temperatures using the gradient function
while Figure 2B shows the comparative result of the same system under
reduced volume. The results showed that each different annealing
temperature gives a different specifc and non-specifc fragment yield,
with highest yield at 65.9 °C while highest specifcity at 70.5 °C. Even
though the difference in each tested temperature is small, the effect was
nonetheless obvious due to the sensitive nature of this PCR system.
These differences hence illustrate the fne control and accuracy the
Mastercycler X50 has over the temperatures across the block.
Additionally, these results were highly reproducible at vastly reduced
reaction volume (3 µL), showing not only that automated pipetting was
very precise, the wells also did not suffer from evaporation problems. At
3 µL, the DNA bands seem to have higher intensity. A possible
explanation is that while the amount of enzyme and DNA were reduced
accordingly in the 3 µL setup, the overall concentration of enzyme and
DNA per total volume is slightly higher, hence resulting in slightly
higher yield. Furthermore, it is expected that the effect caused by
condensation/evaporation in 3 µL is larger than in 10 µL at the same
rate due to the lower amount of water present in the 3 µL setup. This
will thus further driveup the concentration of reagents in 3 µL setup
and may result in slightly higher fnal yield at the same temperatures.
The respective reductions in enzyme consumption and DNA amount required
for successful amplifcation at low reaction volumes have two important
implications. While many inexpensive PCR reagents exist in the market,
DNA polymerases remained one of the most cost-consuming elements in PCR.
This is especially true for users with high-throughput or when more
expensive reagents are needed to increase efciencies when amplifying
difcult targets. Similarly, for users working with precious samples or
low concentration targets, the ability of a thermal cycler to
reproducibly amplify low amounts of samples can be the determining
factor between PCR success or failure.
Conclusion
The results from this study showed that the Eppendorf Mastercycler X50
is a thermal cycler with excellent tem perature performance and
evaporation protection that gives highly reproducible result even for
low volume PCR applications or delicate PCR system.
Combined with the Eppendorf epMotion to handle low volume liquid
handling set-up, a PCR can be set up effortlessly and completed with
minimal time and effort for maximal accuracy and reproducibility.








